The rise in the number of cancer cases across the globe is likely to contribute to the growth of the tumor embolization devices market during the forecast period. According to the American Cancer Society, there were about 1.8 million new cases and 606,880 cancer deaths in 2019 in the USA. The four most common types of cancers worldwide are lung, prostate, bowel, and female breast cancer, accounting for 43% of all the new cancer cases. Therefore, the rise in cancer incidence rate globally is anticipated to boost the demand for the tumor embolization devices market over the forthcoming years.
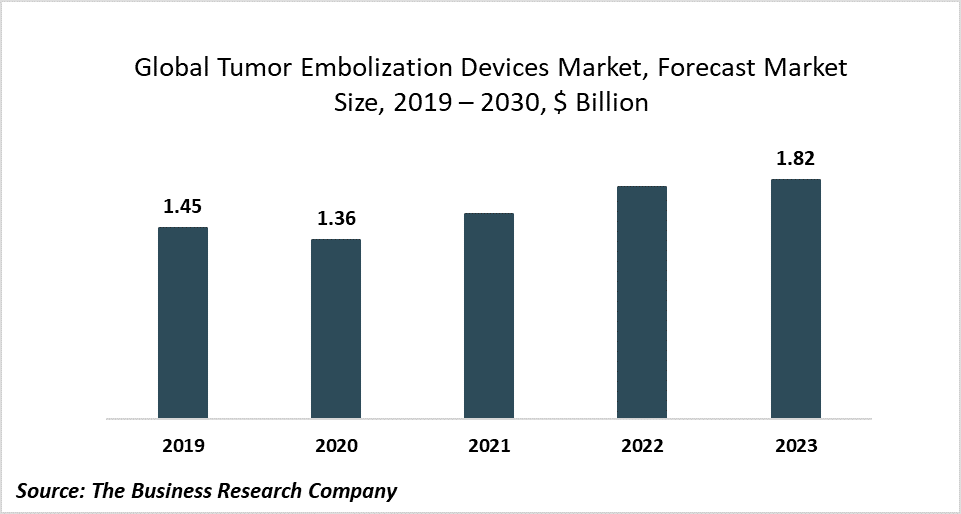
The global tumor embolization devices market is expected to decline from $1.45 billion in 2019 to $1.36 billion in 2020 at a compound annual growth rate (CAGR) of -6.24%. The decline is mainly due to the COVID-19 outbreak that has led to restrictive containment measures involving social distancing, remote working, and the closure of industries and other commercial activities. The entire supply chain has been disrupted, impacting the market negatively. The tumor embolization devices market is then expected to recover and reach $1.82 billion in 2023 at a CAGR of 10.22%.
Companies in the tumor embolization market are increasingly investing in technological advances to provide more efficient devices to the consumers. The advances include the emergence of biocompatible, biodegradable, drug-eluting microspheres that provide effective embolization. Bioresorbable microspheres block the blood vessels causing tumors without long-term blockage of the healthy tissue blood vessels. In 2019, QXMédical announced its decision to license bioresorbable embolic technology from the University of Minnesota, to be used in tumor embolization devices for cancer and tumor treatment.
Regulatory changes are likely to lead to increased costs relating to new product development and service offerings to clients in the tumor embolization devices market. These changes are related to advanced therapies and advanced therapy medicinal products. These regulations include changes to equipment approval procedures and other aspects. For instance, the United States Food and Drug Administration is requesting more clinical data to support claims, therefore increasing the time to process 510(k)s by over 55% over the last decade. The potential loss of revenue due to delays in product release and additional costs incurred due to stringent approval processes puts a strain on investments relating to new product development, thereby affecting the growth of the tumor embolization market.
The tumor embolization devices market consists of sales of tumor embolization devices and related services by entities (organizations, sole traders and partnerships) that manufacture tumor embolization devices. Tumor embolization devices are minimally invasive devices used in the treatment of tumors. These devices are used either to shut down the supply of blood to the tumor or to deliver tumor-killing therapy directly to the tumor.
Here Is A List Of Similar Reports By The Business Research Company:
Interventional Oncology Devices Global Market Report 2020-30: COVID 19 Growth And Change
Tumor Ablation Therapy Devices Global Market Report 2020-30: COVID 19 Growth And Change
Oncology Devices Global Market Report 2020-30: Covid 19 Growth And Change
Oncology Drugs Global Market Report 2020-30: Covid 19 Impact And Recovery
Interested to know more about The Business Research Company?
The Business Research Company is a market intelligence firm that excels in company, market, and consumer research. Located globally it has specialist consultants in a wide range of industries including manufacturing, healthcare, financial services, chemicals, and technology.
Contact Information
The Business Research Company
Europe: +44 207 1930 708
Asia: +91 8897263534
Americas: +1 315 623 0293
Email: info@tbrc.info
Follow us on LinkedIn: https://in.linkedin.com/company/the-business-research-company
Follow us on Twitter: https://twitter.com/tbrc_Info
Media Contact
Company Name: The Business Research Company
Contact Person: Oliver Guirdham
Email: Send Email
Phone: +44 20 7193 0708
Address:
City: London
State: Greater London
Country: United Kingdom
Website: https://www.thebusinessresearchcompany.com/