According to a report published by Grand View Research, Inc.;the global pharmacogenomics technology/theranostics/companion diagnostics (CDx) market is expected to reach a valuation of around USD 18.3 billion by 2025. Rise in demand for cost-effective treatments due to growing pressure on healthcare budgets and innovation are anticipated to drive the market. Growing cost-cutting exercise by payers and government and efforts to improvement advanced products can further fuel the market during the forecast period (2014 to 2025).
Pharmacogenomics and theranostics play a significant role in the development of integrated medicine. Pharmacogenomics testing products can assist physicians in customizing and optimizing therapeutic routine of patients. Growth in this group of specialized molecular diagnostic tests is supporting the real-time assessment of disease treatment. This factor is anticipated to propel adoption in the forthcoming years.
Full Research Report On Pharmacogenomics Technology (Theranostics & CDx) Market Analysis:
www.grandviewresearch.com/industry-analysis/pharmacogenomics-technology-theranostics-companion-diagnostics-cdx-market
Germany pharmacogenomics technology market by therapeutic area, 2014 – 2025 (USD Million)
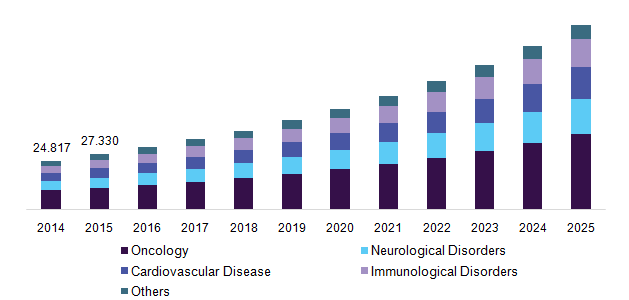
Development of new tests can offer cost-effective treatment and aid in drug-development. Various benefits associated with these diagnostic tests including better therapeutic response monitoring over conventional methods is expected to boost growth of the market for pharmacogenomics. Theranostics’ role in reducing R&D costs as well as time involved in clinical trials can boost its adoption in pharmaceutical companies. Thus, rising pressure on pharmaceutical industry to meet growing demand is likely to fuel market growth in the forthcoming years.
However, challenges regarding early integration of pharmacogenomics-based testing into drug development time lines to get an approval at the same time and issues regarding regulatory policies and reimbursement criteria for the use of companion diagnostic tests are expected to restrict market growth to an extent.
The worldwide pharmacogenomics technology/theranostics/companion diagnostics (CDx) market can be segmented on the basis of therapeutic area, technology, and region. Based on therapeutic area, the market can be segmented into oncology, neurological disorders, cardiovascular disease, immunological disorders, and others. The oncology segment occupied major market share in the past and is expected to provide more opportunities over the forecast period. Theranostics offer cost-effective and precise cancer care. Proper selection of predictive biomarkers, which can provide clinical benefit in a small subset of patients is the key step to implement companion diagnostics in cancer.
Browse More Reports Of This Category By Grand View Research At:
www.grandviewresearch.com/industry/clinical-diagnostics
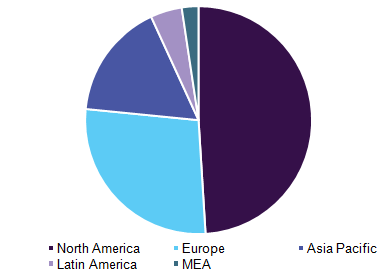
Pharmacogenomics technology (theranostics & CDx) market, by region, 2016 (%)
As per, technology, the market can be categorized into Polymerase Chain Reaction (PCR), in-situ hybridization, immunohistochemistry, sequencing, and others. In 2016, PCR was the major segment in terms of revenue with respect to technology for theranostics. Presence of strong portfolio of assay and various advantages of PCR are anticipated to boost segment growth in near future.
Geographically, the market can be divided into North America, Europe, Asia Pacific, Latin America, and Middle East and Africa.
North America dominated the global market due to presence of established players in the region. Increasing R&D of integrated medicine in U.S. as a result of active participation of government and regulatory bodies is likely to drive regional market growth. The U.S. Food and Drug Administration (FDA) has issued guidance documents to coordinate innovative reviews with different agencies, to define timelines to aid smooth application process, and to clarify suitable regulatory strategy. This factor is expected to bode well for expansion.
Asia Pacific is anticipated to showcase rapid growth during the forecast period on account of advancements in emerging economies. Additionally, increasing demand for pharmacogenomic testing with increasing efforts by players to capitalized on untapped opportunities can have positive impact on regional market over the forecast period.
Leading companies operating in the pharmacogenomics technology/theranostics/companion diagnostics (CDx) market include Thermo Fisher Scientific Inc.; Agilent Technologies; Myriad Genetics Inc.; Thermo Fisher Scientific, Inc.; and AmeriPath Inc. The market has huge opportunities for small players to emphasize on development of novel tests for already licensed drugs. The companies are also collaborating to identify appropriate tumor targets by expansion of identified mutation associated with defective DNA repair. They are also involved in development of single comprehensive tests with capability to capture large amount of relevant content.
Furthermore, market participants are involved in development of theranostics that can target one or two most effective drugs in an individual. They are also involved in development of the assays, which can help understand molecular phenotype.
Grand View Research has segmented the pharmacogenomics technology (theranostics and companion diagnostics) market on the basis of therapeutic area, technology, and region:
Therapeutic Area Outlook (Revenue, USD Million; 2014 – 2025)
-
Oncology
-
Lung Cancer
-
Breast Cancer
-
Colorectal Cancer
-
Cervical Cancer
-
Others
-
-
Neurological Disorders
-
Cardiovascular Disease
-
Immunological Disorders
-
Others
Technology Outlook (Revenue, USD Million; 2014 – 2025)
-
PCR
-
In-situ Hybridization
-
Immunohistochemistry
-
Sequencing
-
Others
Regional Outlook (Revenue, USD Million; 2014 – 2025)
-
North America
-
U.S.
-
Canada
-
-
Europe
-
Germany
-
UK
-
-
Asia Pacific
-
China
-
Japan
-
-
Latin America
-
Brazil
-
-
Middle East & Africa
-
South Africa
-
Explore the BI enabled intuitive market research database, Navigate with Grand View Compass, by Grand View Research, Inc.
About Grand View Research
Grand View Research, Inc. is a U.S. based market research and consulting company, registered in the State of California and headquartered in San Francisco. The company provides syndicated research reports, customized research reports, and consulting services. To help clients make informed business decisions, we offer market intelligence studies ensuring relevant and fact-based research across a range of industries, from technology to chemicals, materials and healthcare.
For more information: www.grandviewresearch.com/
Media Contact
Company Name: Grand View Research, Inc.
Contact Person: Sherry James, Corporate Sales Specialist – U.S.A.
Email: Send Email
Phone: 1-415-349-0058, Toll Free: 1-888-202-9519
Address:28 2nd Street, Suite 3036
City: San Francisco
State: California
Country: United States
Website: www.grandviewresearch.com/industry-analysis/pharmacogenomics-technology-theranostics-companion-diagnostics-cdx-market

