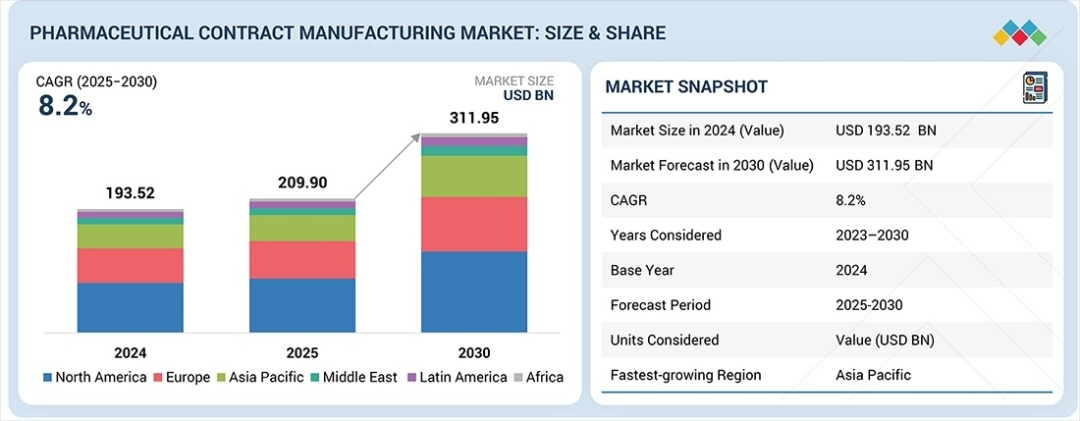
The global pharmaceutical contract manufacturing market is entering a phase of unprecedented growth, projected to surge from USD 209.90 billion in 2025 to USD 311.95 billion by 2030. This expansion, representing a compound annual growth rate (CAGR) of 8.2%, is fundamentally reshaped by a “perfect storm” of industry shifts. Key drivers include a critical capacity crunch for GLP-1 agonist drugs, a rapid increase in Antibody-Drug Conjugate (ADC) development, and the imminent loss of patent exclusivity for several blockbuster biologics. As big pharmaceutical companies face mounting pricing pressures and the need for faster speed-to-market, the reliance on Contract Development and Manufacturing Organizations (CDMOs) has transitioned from a tactical cost-saving measure to a core strategic necessity.
Download PDF Brochure:https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=201524381
Technological Transformation and the Biologics Evolution
The pharmaceutical manufacturing landscape is undergoing a radical technical overhaul to accommodate the increasing complexity of modern medicine. The industry is rapidly moving away from traditional batch processing toward continuous manufacturing and single-use bioprocessing systems. These technologies allow CDMOs to offer greater scalability and reduced cross-contamination risks, which are essential for the production of highly sensitive large molecules. As of 2024, the large molecule segment has emerged as the dominant force in the market, a trend expected to accelerate through 2030. This dominance is fueled by the explosion of the biologics pipeline, including monoclonal antibodies and recombinant proteins, which require specialized GMP facilities and high-containment environments that many pharmaceutical companies prefer to outsource rather than build in-house.
Furthermore, the rise of Cell and Gene Therapies (CGT) has opened a high-value frontier for contract manufacturers. These therapies require hyper-specialized viral vector manufacturing and automated fill-finish systems. Leading players like Thermo Fisher Scientific Inc, Lonza, and Catalent, Inc. are aggressively investing in these capabilities to meet the stringent biosafety standards required. This technological arms race ensures that CDMOs are no longer just “hired hands” but are instead vital innovation partners capable of handling the most complex molecular formats in the history of medicine, ranging from mRNA vaccines to personalized oncology treatments.
Strategic Outsourcing Dynamics and the GLP-1 Impact
One of the most significant immediate disruptions in the market is the GLP-1 capacity crunch. With the global surge in demand for weight-loss and diabetes medications, pharmaceutical innovators are facing severe bottlenecks in peptide synthesis and sterile injectable filling. This has resulted in a massive influx of secondary outsourcing contracts, as companies scramble to secure guaranteed production lines. CDMOs are responding by expanding their “one-stop-shop” models, providing end-to-end services from drug substance manufacturing to final packaging and labeling. This model is particularly attractive to big pharmaceutical companies, which currently dominate the end-user segment and are projected to grow at a CAGR of 8.7% as they seek to streamline their global supply chains and mitigate the risks associated with multi-vendor management.
The market is also reacting to the “patent cliff” affecting several high-value biologics. As original patents expire, the demand for biosimilars is skyrocketing, particularly in emerging markets. This provides a dual opportunity for contract manufacturers: they can partner with innovators to optimize the lifecycle of a drug through more cost-effective production, or they can work with generic manufacturers to bring affordable biosimilars to market rapidly. This shift is particularly evident in the Asia Pacific region, which is slated to be the fastest-growing geographical market. Countries like India, China, and South Korea are leveraging lower labor costs and significant government investments in GMP-certified infrastructure to position themselves as the world’s primary hubs for large-scale pharmaceutical and biologics production.
Request Sample Pages-https://www.marketsandmarkets.com/requestsampleNew.asp?id=201524381
Navigating Global Challenges and Supply Chain Resilience
Despite the robust growth projections, the pharmaceutical contract manufacturing market faces a complex array of challenges, most notably global trade instability and the rising trend of insourcing for critical components. Geopolitical tensions and fluctuating tariffs have forced many CDMOs to reassess their sourcing strategies for Active Pharmaceutical Ingredients (APIs) and raw materials. To combat this, many “Star” players are diversifying their regional footprints, establishing “local-for-local” manufacturing sites in North America and Europe to ensure supply chain resilience against future global disruptions. This move toward regionalization is a direct response to the need for data integrity, quality consistency, and the mitigation of logistics bottlenecks that were exposed during previous global health crises.
Moreover, the industry is grappling with pricing pressures on innovator drugs, driven by government reimbursement tightening and the increased penetration of generics. This economic environment forces pharmaceutical companies to lean even harder on CDMOs for operational efficiency. To stay competitive, contract manufacturers are integrating Artificial Intelligence (AI) and Machine Learning into their quality control and process analytical technology (PAT). These digital disruptions allow for real-time monitoring of batch reliability and faster scale-up, significantly reducing the cost of failure. As the market moves toward 2030, the ability of a CDMO to provide not just capacity, but also high-level digital integration and regulatory expertise, will be the primary differentiator in securing long-term, high-value partnerships with the world’s leading life sciences companies.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com