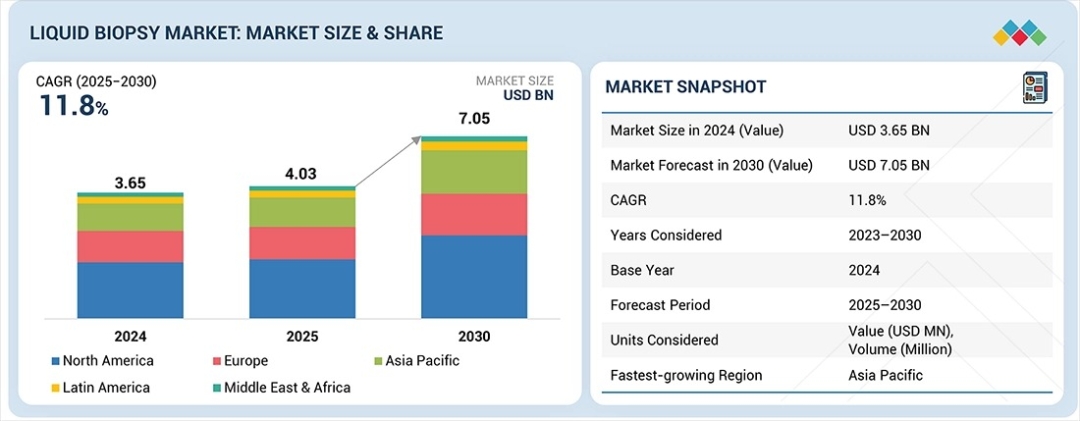
The liquid biopsy market is experiencing exceptional growth, valued at USD 4.03 billion in 2025 and projected to expand to USD 7.05 billion by 2030, representing a commanding compound annual growth rate (CAGR) of 11.8% throughout the forecast period. This acceleration reflects a fundamental transformation in oncology care, where precision diagnostics are becoming central to treatment decision-making and patient outcomes for healthcare organizations worldwide. The escalating global cancer burden represents the primary catalyst for liquid biopsy adoption, as cancer incidence and prevalence continue to rise across developed and emerging markets. Healthcare systems are investing in diagnostic solutions that enable early detection, treatment optimization, and disease progression monitoring. Liquid biopsy delivers critical advantages over traditional tissue biopsies—it is minimally invasive, delivers faster results, and enables real-time disease tracking without patient discomfort or procedural complications.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=13966350
Why Early Cancer Detection Is Reshaping Clinical Strategy
Leading health organizations worldwide are emphasizing cancer screening initiatives, recognizing that early detection substantially improves treatment efficacy and patient survival rates. Liquid biopsy addresses this imperative by enabling detection of circulating tumor DNA (ctDNA), circulating tumor cells (CTCs), and other actionable biomarkers through a simple blood draw, making repeated monitoring practical and cost-effective compared to invasive tissue biopsy procedures. Early cancer detection is becoming a strategic priority because it allows clinicians to identify tumors at more treatable stages, reducing treatment complexity and improving outcomes. The rising adoption of preventive healthcare and precision medicine initiatives is accelerating the integration of liquid biopsy into routine oncology practice, positioning it as an essential diagnostic tool across hospitals, reference laboratories, and academic research centers.
How Clinical Applications Are Driving Market Expansion
Liquid biopsy addresses four critical clinical scenarios: early cancer screening, therapy selection, treatment monitoring, and recurrence surveillance. Therapy selection represents the largest application segment, driven by liquid biopsy’s ability to detect actionable genetic alterations that guide personalized treatment decisions. By identifying which patients will respond to specific targeted therapies, clinicians can match patients with optimal treatment regimens, enhancing efficacy and reducing unnecessary side effects. Collaboration between pharmaceutical and diagnostic companies is fostering co-development of targeted therapies and companion diagnostic tests, creating a virtuous cycle that reinforces clinical adoption. The rising adoption of precision medicine and companion diagnostics is accelerating integration of liquid biopsy into routine oncology practice, making it central to modern cancer management strategies.
Next-Generation Sequencing Powers Diagnostic Innovation
Next-generation sequencing (NGS)-based multi-gene analysis is rapidly becoming the preferred technology platform, enabling simultaneous detection of multiple genetic alterations with high accuracy. Unlike single-gene PCR and microarray approaches, NGS provides comprehensive molecular profiling essential for identifying complex genomic changes that drive therapy selection and treatment response monitoring. Its high-throughput nature and adaptability make it uniquely suited for personalized oncology applications, positioning it as the foundation of precision cancer care. Circulating tumor DNA (ctDNA) is emerging as the dominant biomarker due to its exceptional utility in cancer detection, treatment monitoring, and minimal residual disease identification. A single blood draw provides detailed genetic insights with minimal patient burden, making ctDNA a practical and reliable alternative to invasive tissue biopsies. Its ability to track tumor evolution in real time also enables clinicians to detect treatment resistance early and adjust therapy accordingly, driving superior patient outcomes.
Assay Kits and Blood-Based Sampling Lead Product Adoption
Assay kits continue to dominate product and service revenue, driven by their essential role in routine testing and frequent replenishment cycles. As liquid biopsy adoption expands across oncology and emerging clinical applications, the demand for highly specific and sensitive assays continues to strengthen. The growing need for reliable testing across early cancer detection, treatment monitoring, and companion diagnostics reinforces this segment’s trajectory. Blood-based sampling represents the primary sample type, leveraging its critical advantages: ease of collection, minimal invasiveness, and comprehensive biomarker information. Blood-based liquid biopsy is safer, faster, and more patient-acceptable than traditional tissue biopsies, driving broad adoption across diagnostic laboratories, hospitals, and research institutions. Ongoing technological advancements and expanding regulatory support position blood as the preferred specimen type for cancer diagnostics.
Reference Laboratories Anchor Market Growth Across Healthcare Systems
Reference laboratories serve as the primary market drivers, offering high testing volumes, advanced infrastructure, and specialized expertise in molecular analysis. These facilities play a crucial role in supporting oncology diagnostics, companion diagnostics, and clinical research applications. Hospitals, physician laboratories, and academic research centers provide decentralized testing capabilities and specialized services that expand access to liquid biopsy diagnostics. North America maintains the largest market share, supported by advanced healthcare infrastructure, strong research and development capabilities, and significant investment in cancer diagnostics. Leading diagnostic companies, robust regulatory frameworks, and widespread awareness of early detection benefits are driving rapid adoption across hospitals, reference laboratories, and academic institutions throughout the region.
Diagnostic Sensitivity and Reimbursement Present Key Market Challenges
Certain liquid biopsy procedures demonstrate lower sensitivity and specificity, particularly in detecting circulating tumor DNA (ctDNA) at extremely low concentrations. This limitation can result in false-negative findings, delaying tumor detection in early-stage cancers or minimal residual disease monitoring. Statistical and sampling challenges exacerbate this issue, potentially impacting treatment decisions and patient outcomes. Healthcare organizations must carefully evaluate technology platforms and validation methodologies to ensure diagnostic reliability. The complex and inconsistent reimbursement landscape presents a significant barrier to liquid biopsy adoption. High-cost molecular tests frequently face unclear coverage policies, and patients often encounter unexpected costs post-billing. While some payers cover comprehensive multi-gene panels, coverage is sometimes restricted to single-gene analysis, limiting clinical utility. Healthcare providers must proactively engage with payers and advocate for transparent reimbursement policies that ensure patient access to life-saving diagnostics.
Companion Diagnostics Create Transformative Market Opportunities
Companion diagnostics represent a transformative opportunity, as precision medicine expands the role of biomarker-guided therapy selection. Liquid biopsy’s non-invasive capability to detect actionable biomarkers positions it as the ideal diagnostic modality for identifying patients most likely to benefit from specific targeted therapies. This strategic intersection of diagnostics and therapeutics is accelerating pharmaceutical-diagnostic collaborations and driving exponential adoption across healthcare systems globally. The growing importance of companion diagnostics in oncology care is fostering closer partnerships between pharmaceutical manufacturers and diagnostic companies, enabling the simultaneous development of drugs and their associated diagnostic tests. This integrated approach enhances treatment outcomes and creates sustainable competitive advantages for organizations investing in precision oncology infrastructure.
Request Sample Pages: https://www.marketsandmarkets.com/requestsampleNew.asp?id=13966350
Industry Leaders Drive Innovation and Market Transformation
Global innovation is being driven by established diagnostic leaders including Natera, Inc. (United States), Guardant Health (United States), Myriad Genetics, Inc. (United States), Illumina, Inc. (United States), F. Hoffmann-La Roche Ltd. (Switzerland), QIAGEN (Netherlands), Exact Sciences Corporation (United States), Thermo Fisher Scientific Inc. (United States), Sysmex Corporation (Japan), and Bio-Rad Laboratories, Inc. (United States). These organizations continue advancing liquid biopsy technologies, expanding clinical evidence, and establishing industry standards that shape precision oncology practice. Their investments in research and development, regulatory compliance, and clinical validation are accelerating market adoption and establishing liquid biopsy as the standard of care in modern cancer management.
Strategic Imperative for C-Suite Executives in Healthcare and Diagnostics
The 11.8% CAGR expansion through 2030 represents both a clinical imperative and a compelling strategic opportunity. Healthcare systems, diagnostic companies, and oncology providers that strategically invest in liquid biopsy infrastructure and precision oncology capabilities will strengthen competitive positioning, improve patient outcomes, and capture market share in the rapidly evolving cancer diagnostics landscape. For C-suite executives, this represents the convergence of clinical necessity, patient demand for minimally invasive diagnostics, and robust market tailwinds. Organizations that align capital investment with precision oncology strategy and position themselves to adopt liquid biopsy technologies will lead in cancer care delivery and gain sustainable competitive advantage. The time to act is now—as liquid biopsy transforms from an emerging technology into a standard diagnostic tool, early adopters will establish market leadership and drive superior financial performance while fundamentally improving patient outcomes across oncology care.
Related Reports:
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com/Market-Reports/liquid-biopsy-market-13966350.html