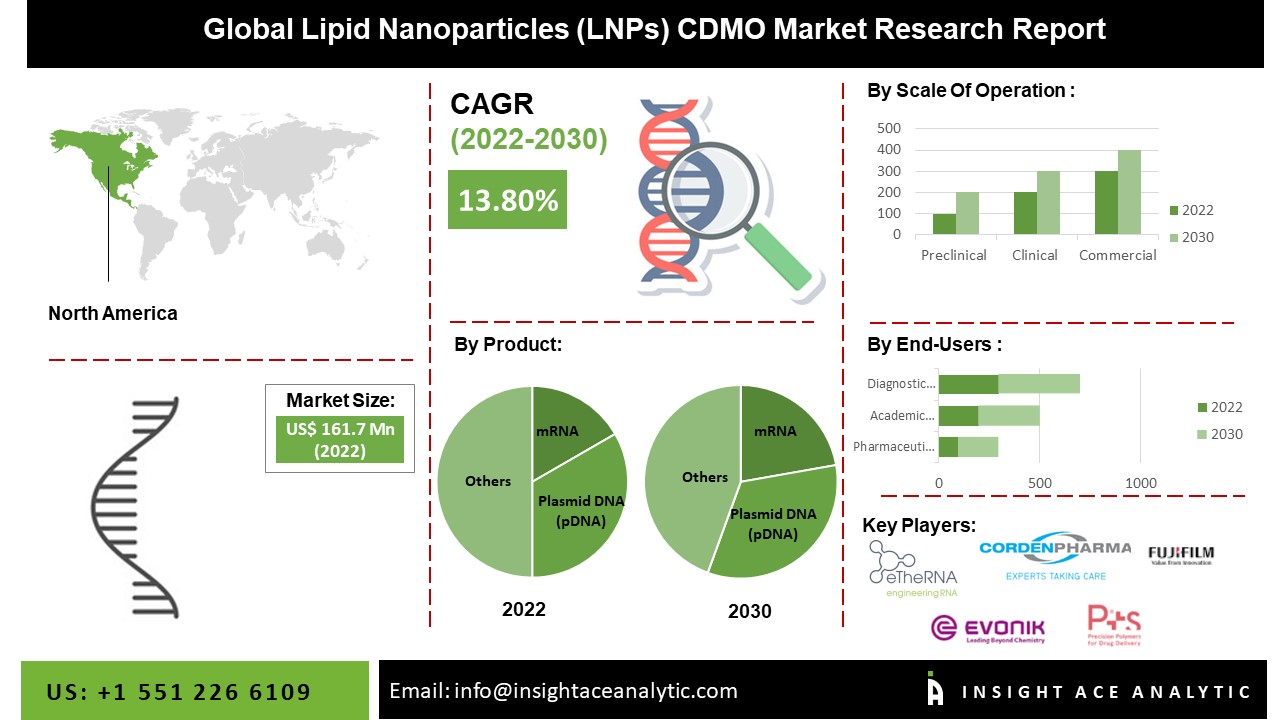
“The Lipid Nanoparticles (LNPs) CDMO Market” in terms of revenue was estimated to be worth $182.0 Mn in 2023 and is poised to reach $518.2 Mn by 2031, growing at a CAGR of 14.10% from 2024 to 2031 according to a new report by Insight Ace Analytic.
Request for Sample Pages: https://www.insightaceanalytic.com/request-sample/1432
Latest Drivers Restraint and Opportunities Market Snapshot:
Key factors influencing the global Lipid Nanoparticles (LNPs) CDMO Market are:
- The rising chronic and infectious diseases
- Personalized medicines
- An increasing number of health-conscious people
The following are the primary obstacles to the Lipid Nanoparticles (LNPs) CDMO market’s expansion:
- High-cost outsourced services
- Lack of specialized expertise in nanoparticles production
- Stringent rules and regulations
Future expansion opportunities for the global The Lipid Nanoparticles (LNPs) CDMO market include:
- Rising collaboration for lipid nanoparticle manufacturing
- Oncology applications
- Adoption of single-use technology
Market Analysis:
One of major driving factors of the Lipid Nanoparticles (LNPs) CDMO market is the surging medical applications of nanoparticles. An increase in investments by key players in lipid nanoparticles to develop promising drug therapies, rising cases of chronic diseases, an increasing aging population, and a growing number of health-conscious people also contribute to the market’s growth.
List of Prominent Players in the Lipid Nanoparticles (LNPs) CDMO Market:
- Laboratorios Farmacéuticos Rovi, S.A.
- Samsung Biologics
- Thermo Fisher Scientific
- Sartorius AG (BIA Separations)
- AGC Biologics
- Hanmi Pharmaceutical
- BioCina Pty Ltd.
- Catalent, Inc.
- Genevant Sciences
- Lonza Group AG
- Rentschler Biopharma
- Nitto Denko Avecia
- Evonik
- Orden Pharma GmbH
- eTheRNA
- Polypeptide Therapeutic Solutions(PTS)
- FUJIFILM Corporation
- ST Pharm Co Ltd.
- Exalead (Merck KGaA)
- Avanti Polar Lipids, Inc. (Croda International Plc.
- Emergent CDMO
- Esco Aster Pte Ltd
- Ernal Biosciences
- Recipharm AB
- Phosphorex Inc.
- Polymun Scientific Immunbiologische Forschung GmbH
- BIOVECTRA
- Vernal Biosciences
- Helix Biotech
Lipid Nanoparticles (LNPs) CDMO Market Report Scope:
|
Report Attribute |
Specifications |
|
Market size value in 2023 |
USD 182.0 Mn |
|
Revenue forecast in 2031 |
USD 518.2 Mn |
|
Growth rate CAGR |
CAGR of 14.10% from 2024 to 2031 |
|
Quantitative units |
Representation of revenue in US$ Million, and CAGR from 2024 to 2031 |
|
Historic Year |
2019 to 2023 |
|
Forecast Year |
2024-2031 |
|
Report coverage |
The forecast of revenue, the position of the company, the competitive market structure, growth prospects, and trends |
|
Segments covered |
By Product, Scale of Operation, End-User |
|
Regional scope |
North America; Europe; Asia Pacific; Latin America; Middle East & Africa |
Recent Developments:
- In May 2022, ST Pharm launched its global mRNA Consignment Development and Manufacturing Organization (CDMO) business by signing a lipid supply agreement with Biotech in North America. Lipids play a crucial role in the production of mRNA-LNPs.
- In Mar 2022, eTheRNA Manufacturing introduced the novel LNP formulation development and production services. The new LNP service allows for precise delivery and personalized distribution of substances by utilizing eTheRNA’s unique lipid libraries and formulations and also allows customers to optimize the delivery of their RNA products by leveraging the expertise of its specialized team.
- In July 2021, Curia (US), formerly AMRI, a contract research, development, & manufacturing organization, acquired Integrity Bio, Inc., a biopharmaceutical & fill-finish organization in Camarillo, to enhance its biologics drug product formulation development and fill-finish network.
Curious about this latest version of the report? @ https://www.insightaceanalytic.com/enquiry-before-buying/1432
The Lipid Nanoparticles (LNPs) CDMO Market Dynamics:
Market Drivers: Personalized Medicines
Personalized medicines are essential for delivering drugs at specifically targeted sites. Liposomes, niosomes, nanoparticles, and nanotechnology-based drug delivery systems are helpful in several therapeutic areas. It is projected to be effective in a subset of patients, leaving others with either ineffective treatment or treatment that causes significant toxicity. Furthermore, Lipid nanoparticles are used as a carrier in delivering oncology and neurology-related drugs. It is anticipated to individualize/customize therapeutic management based on the patient’s characteristics to overcome blanket treatment.
Challenges: Stringent Rules and Regulations
The stringent rules and regulations imposed on the industries to make and market drugs have restricted them from growing. The laws issued by several governments also restricted the inflow of R&D investments for developing complex drugs using nanoparticles. Furthermore, stringent regulatory necessities for drug approval and safety testing pose essential barriers to Lipid Nanoparticles development and commercialization. Meeting regulatory values takes time and effort, restraining the growth of the industry.
North America Is Expected To Grow With the Highest CAGR during the Forecast Period
The North America the Lipid Nanoparticles (LNPs) CDMO Market is likely to register a significant revenue share and develop at a rapid CAGR soon. This is due to the increasing awareness about the applications of nanoparticles, increasing government interest and investments in research and developments. The increasing prevalence of chronic diseases drives the North American market. Furthermore, the region is expected to dominate the market owing to the well-established healthcare facilities & CMOS, the rising demand for specialized drugs, and the increasing number of clinical trial advancements in the biopharmaceutical industries.
Segmentation of the Lipid Nanoparticles (LNPs) CDMO Market-
By Product-
- mRNA
- Plasmid DNA (pDNA)
- siRNA
- saRNA
- microRNA
- Others
By Scale of Operation
- Preclinical Scale Operations
- Clinical Scale Operations
- Commercial Scale Operations
By End-User-
- Pharmaceuticals Companies
- Academic Research Institutes
- Diagnostic Laboratories
By Region-
North America-
- The US
- Canada
- Mexico
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of Middle East and Africa
For More Customization @ https://www.insightaceanalytic.com/customisation/1432
Media Contact
Company Name: InsightAce Analytic Pvt. Ltd
Contact Person: Diana D’Souza
Email: Send Email
Country: United States
Website: https://www.insightaceanalytic.com/