The availability of private and government funding drove the interventional oncology devices market. In the field of cancer, funding from the government and private organizations has increased, especially for the devices used in treatments, as it promotes early detection and better survival rates. The American Cancer Society, the largest non-government, not-for-profit cancer research funder, awarded 93 grants which totaled $40 million in the first of two cycles for 2019 cancer research. In 2019, the US Congress provided NCI with an amount of $5.74 billion, which represents a $79 million increase from FY 2018, and an additional $400 million for the Cancer Moonshot program that has a special focus on early detection of cancer using latest technologies. BioProtect Ltd, an Israeli company that develops novel bioabsorbable polymer spacer balloons, received a funding of $13 million of its Series D equity financing from a leading global syndicate of venture investors that finances the ongoing multicenter FDA clinical trial of BioProtect’s lead product, the ProSpace balloon spacer, which protects prostate cancer patients undergoing radiation therapy. Endosight, the first augmented-reality guided ablative system, received 20% of its funding from private organizations. The funding from government and private organizations help in the development of new technologies and devices, thereby driving the interventional oncology medical devices market.
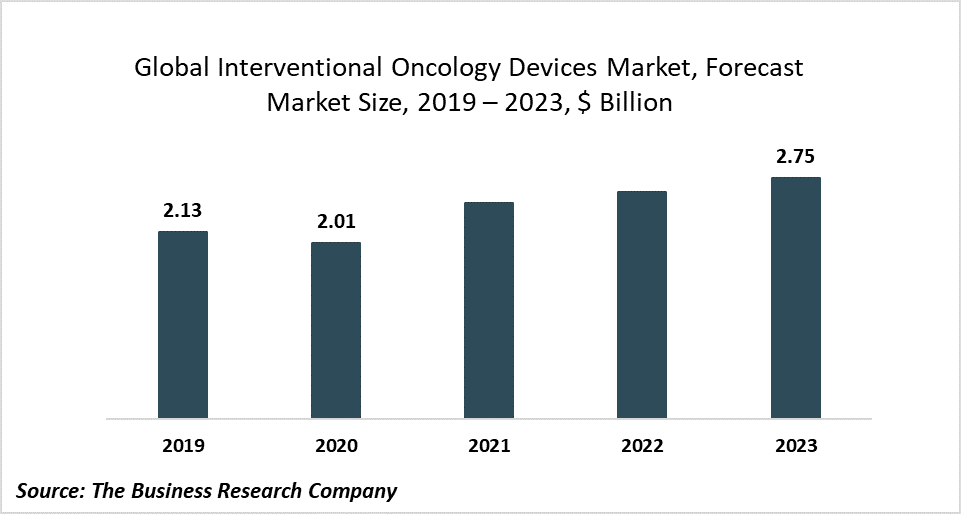
The global interventional oncology device market is expected to decline from $2.13 billion in 2019 to $2.01 billion in 2020 at a compound annual growth rate (CAGR) of -5.39%. The decline is mainly due to the COVID-19 outbreak that has led to restrictive containment measures involving social distancing, remote working, and the closure of industries and other commercial activities. The entire supply chain has been disrupted, impacting the market negatively. The interventional oncology medical devices market is then expected to recover and reach $2.75 billion in 2023 at a CAGR of 10.99%.
Companies in the market are focusing on deploying robotics technology in interventional oncology devices for higher efficiency. The robotic system integrates image-based planning and navigation with the installation of different instruments to the desired body part with better accuracy and efficiency. For instance, in November 2019, the first robotic device by XACT Robotics Ltd was approved in the United States by the Food and Drug Administration for use during computed tomography (CT) controlled percutaneous interventional procedures. Similarly, in March 2018, Auris Surgical Robotics, Inc received FDA clearance for the Monarch™ Platform, a lung biopsy platform that uses robotic technology.
The market for interventional oncology devices comprises the sale of interventional oncology devices and related services by the entities that manufacture them. Interventional oncology devices are used to detect and treat cancer using minimally invasive procedures and advanced imaging technologies.
Here Is A List Of Similar Reports By The Business Research Company:
Oncology Devices Global Market Report 2020-30: Covid 19 Growth And Change
Tumor Embolization Devices Global Market Report 2020-30: COVID 19 Growth And Change
Interventional Cardiology Devices And Equipment Global Market Report 2020
Interventional Neurology Devices And Equipment Global Market Report 2020
Oncology Drugs Global Market Report 2020-30: Covid 19 Impact And Recovery
Media Contact
Company Name: The Business Research Company
Contact Person: Oliver Guirdham
Email: Send Email
Phone: +44 20 7193 0708
Address:
City: London
State: Greater London
Country: United Kingdom
Website: https://www.thebusinessresearchcompany.com/