InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “Global In Vitro Diagnostic (IVD) Reagents Market – (By Type (Antibodies, Purified Proteins and Peptides, Oligonucleotides, Others), By Technology (Immunoassay, Clinical Chemistry, Molecular Diagnostics, Microbiology, Hematology, Others), By Use (Clinical Use, Research Use, Others), By End User (IVD Manufacturers, Academic Institutes & Research Laboratories, Hospital Laboratories, Others)), Trends, Industry Competition Analysis, Revenue and Forecast To 2031.”
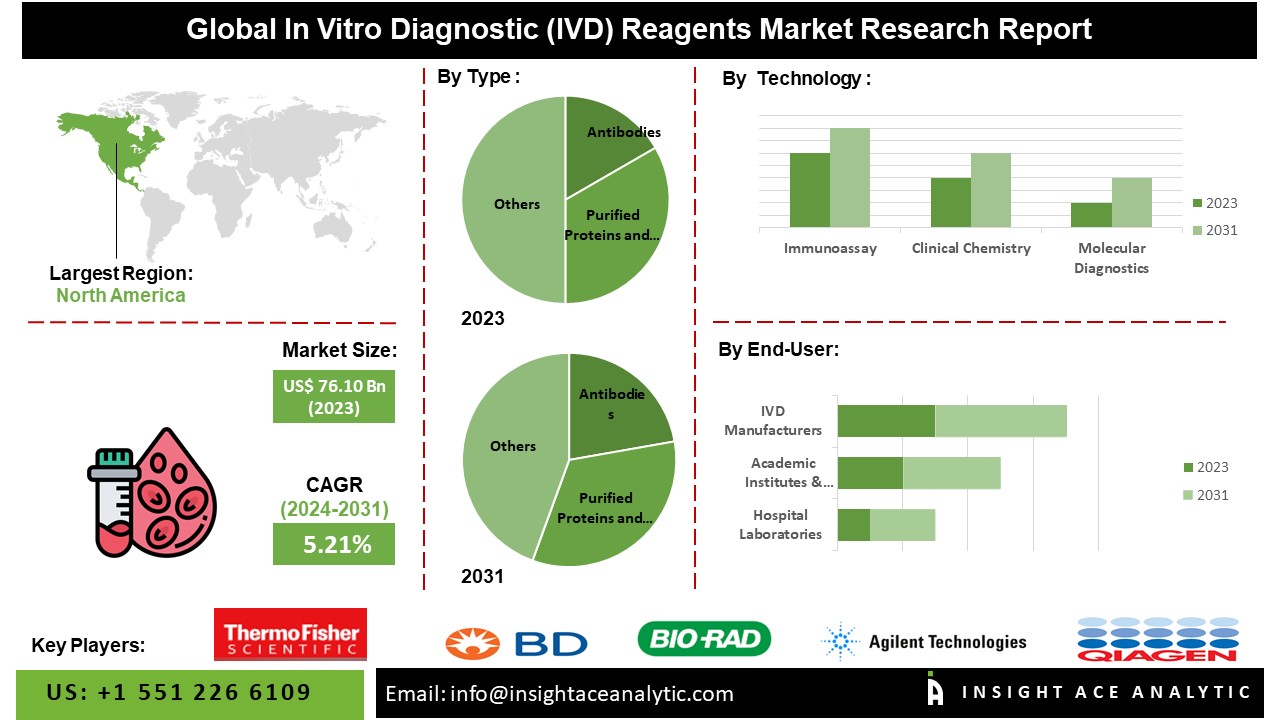
According to the latest research by InsightAce Analytic, the Global In Vitro Diagnostic (IVD) Reagents Market is valued at US$ 76.10 Billion in 2023, and it is expected to reach US$ 113.15 Billion by 2031, with a CAGR of 5.21 % during the forecast period of 2024-2031.
In vitro diagnostic (IVD) reagents are essential components of medical tests that aid in the detection of diseases and other health concerns, allowing for the improvement of treatment decisions. They help monitor the patient’s vitals and are utilized for precise diagnostic testing of illnesses and other underlying conditions. Each patient’s best course of treatment can be better determined with the help of these diagnostic tests. With the rise in both acute and chronic disorders continuing to rise, the in vitro diagnostics business is seeing rapid growth. The frequency of long-term health problems, such as diabetes and cancer, heart disease, and tuberculosis, is on the rise in modern workplaces, which is cause for concern. There has been a staggering rise in the number of cases of infectious infections, respiratory disorders, and gastrointestinal difficulties. It is anticipated that the demand for diagnostic equipment will be driven by the increasing occurrence of these illnesses, which drives the IVD market. The rise in investments in research and development (R&D) aimed at developing more affordable treatments is likely to open up promising prospects for market growth. However, the high cost of instruments and the maintenance of diagnostic tablets impeded the market’s growth.
Request for Sample Pages: https://www.insightaceanalytic.com/request-sample/2452
List of Prominent Players in the In Vitro Diagnostic (IVD) Reagents Market:
- Thermo Fisher Scientific Inc
- Becton, Dickinson and Company
- Bio-Rad Laboratories, Inc
- Agilent Technologies, Inc.
- QIAGEN N.V.
- Merck KGaA
- Beckman Coulter, Inc
- Hologic, Inc
- Abcam Ltd
- Bio-Techne Corp
- InBios International, Inc.
- SDIX, LLC
- RayBiotech, Inc.
- Sino Biological Inc.
- Other Market Players
Market Dynamics:
Drivers-
The growing demand for the in vitro diagnostic (IVD) reagents market is fueled by many reasons that underscore the market’s significance in contemporary healthcare. The prevalence of long-term health problems is increasing, which is a key factor propelling the IVD Reagents Market. Diabetes, heart disease, cancer, and infectious disorders like hepatitis and HIV are all on the increase. When it comes to diagnosing these disorders, intravenous diagnostic reagents are vital. They allow for early identification, disease monitoring, and therapy assessment. Intravenous diagnostic reagents are in high demand due to the rising prevalence of chronic disorders. Advances in diagnostic technology have boosted patient outcomes, which has fueled market expansion.
Challenges:
The prime challenges are high cost, a shortage of competent individuals, and a lack of norms and protocol because of lockdowns and isolation in emerging countries, which are predicted to slow the growth of the in vitro diagnostic (IVD) reagents market. Healthcare systems around the world are dealing with limited funding, which forces providers to find ways to save costs without sacrificing quality. Under these conditions, there is a lot of pressure on IVD reagent producers to lower production costs without sacrificing quality or dependability. Healthcare providers and consumers are both price-sensitive, which makes the problem even worse. The COVID-19 pandemic broke out, and virus-related disruptions have become increasingly widespread in several industries, including the worldwide in-vitro diagnostic market. Service providers have prioritized sanitizing and reorganizing workspaces as a defence mechanism to increase resilience and decrease security risks. Integrating digital collaboration tools has also been a need for remote customer support. Businesses in the in-vitro diagnostic industry are gradually making a return from the recession by embracing digital technologies that allow them to grow their customer base and increase productivity.
Regional Trends:
The North American in vitro diagnostic (IVD) reagents market is anticipated to record a maximum market revenue share. It is projected to grow at a high CAGR in the near future because local businesses have a strong presence in the area, and the infrastructure supporting various industries is expanding. The rising prevalence of long-term health issues, including diabetes, heart disease, and cancer, is also a key factor propelling the IVD industry forward. Besides, Asia Pacific had a considerable share in the market due to improvements in healthcare expenditure and increased usage of point-of-care devices.
Curious About This Latest Version Of The Report? Enquiry Before Buying: https://www.insightaceanalytic.com/report/in-vitro-diagnostic-ivd-reagents-market/2452
Recent Developments:
- In April 2023, ALPCO-GeneProof and Thermo Fisher formed a partnership to expand the former’s portfolio of CE-IVD molecular assays. Thermo Fisher provided a portfolio comprising 37 CE-IVD molecular diagnostic assays that were originally included in the portfolio of ALPCO-GeneProof. The assays were constructed utilizing their groundbreaking “one workflow” technology, which facilitated usability and ensured compatibility with an extensive array of qPCR instruments.
- In June 2022, Biotec CerTest and BD Jointly developed a molecular diagnostic assay for monkeypox. The collaboration involved the utilization of the BD MAX™ open system reagent suite in the assay to verify the functionality of the CerTest VIASURE Monkeypox CE/IVD molecular test on the BD MAX™ System. The assay, which was accessible to BD MAX™ users, had the potential to contribute to the advancement of knowledge regarding the worldwide dissemination of the disease.
Segmentation of In Vitro Diagnostic (IVD) Reagents Market-
By Type-
- Antibodies
- Purified Proteins and Peptides
- Oligonucleotides
- Others
By Technology-
- Immunoassay
- Clinical Chemistry
- Molecular Diagnostics
- Microbiology
- Hematology
- Others
By Use-
- Clinical Use
- Research Use
- Others
By End user-
- IVD Manufacturers
- Academic Institutes & Research Laboratories
- Hospital Laboratories
- Others
By Region-
North America-
- The US
- Canada
- Mexico
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- South East Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
For More Customization @ https://www.insightaceanalytic.com/customisation/2452
Media Contact
Company Name: InsightAce Analytic Pvt. Ltd
Contact Person: Diana D’Souza
Email: Send Email
Country: United States
Website: https://www.insightaceanalytic.com/