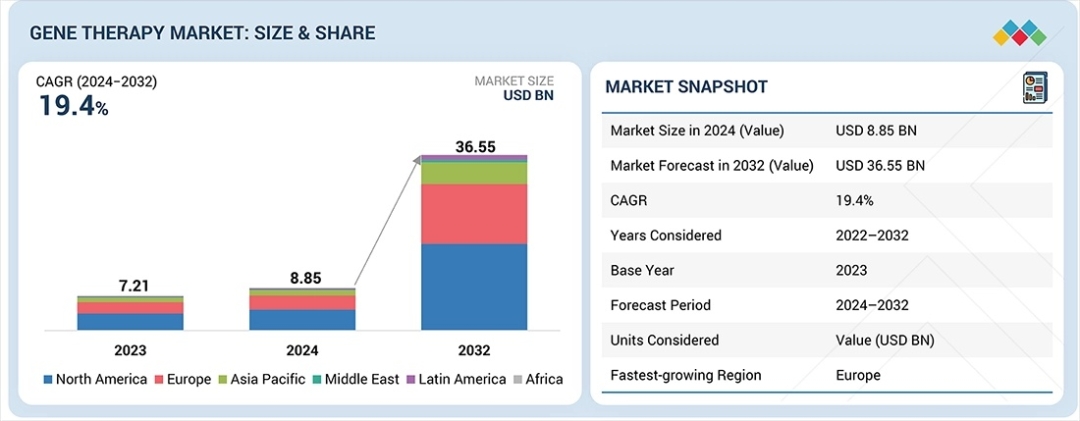
The global Gene Therapy Market is entering a transformative era of growth, with its valuation projected to surge from USD 8.85 billion in 2024 to USD 36.55 billion by 2032. According to the latest analysis by MarketsandMarkets, the industry is set to expand at a robust CAGR of 19.4% over the forecast period. This rapid escalation is primarily fueled by a paradigm shift from symptom management to curative interventions, particularly for genetic disorders, rare diseases, and oncology. As of 2024, North America continues to lead the global landscape with a 49.4% market share, supported by an intensive research and development environment. Key market drivers include the increasing volume of regulatory approvals for orphan drugs, significant venture capital investments, and a robust clinical pipeline that promises to redefine the standard of care for chronic conditions.
Download PDF Brochure:https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=122857962
The Neurological Frontier and the Rise of Curative Precision Medicine
The neurology segment has emerged as a powerhouse within the gene therapy ecosystem, projected to grow at the fastest rate through 2032. This dominance is underscored by the rising incidence of neurodegenerative and neuromuscular disorders, such as Spinal Muscular Atrophy (SMA), ALS, and Parkinson’s disease. Innovative therapies like Zolgensma by Novartis AG have set a precedent for the industry, demonstrating the profound impact of delivering functional gene copies to motor neurons. Recent breakthroughs, such as the FDA’s accelerated approval of PTC Therapeutics’ Kebilidi the first-ever gene therapy administered directly to the brain highlight the sector’s move toward highly localized and effective delivery methods. This trend toward precision medicine is allowing clinicians to address the root genetic causes of brain-related ailments that were previously considered untreatable. Furthermore, the increasing focus on the “deficiency” gene type segment reinforces this growth, as more therapies are developed to replace or supplement missing genetic material, offering a one-time curative potential that traditional pharmaceuticals cannot match.
Strategic Advancements in Viral Vector Engineering and Delivery Infrastructure
Technological evolution in delivery mechanisms remains a cornerstone of the market’s expansion. In 2024, the viral vector segment dominated the market, with Adeno-associated virus (AAV) and lentiviral vectors serving as the primary vehicles for gene transport. The industry is witnessing a significant trend in capsid engineering; for instance, Novartis AG’s recent acquisition of Kate Therapeutics for USD 1.1 billion granted them access to advanced platforms that “evolve” AAV capsids for higher potency. Simultaneously, the In Vivo delivery method maintains a commanding 72.5% market share, largely because it allows for direct internal modification without the complexity of external cell manipulation. The intravenous (IV) route of administration is also seeing a high CAGR of 19.9%, favored for its non-invasive nature and ability to achieve systemic distribution of therapeutic material. These advancements are supported by a maturing infrastructure within hospitals and specialty centers, which are increasingly equipped with the specialized laboratories and surgical units required to manage the high-stakes administration and monitoring of these complex biological products.
Request Sample Pages-https://www.marketsandmarkets.com/requestsampleNew.asp?id=122857962
Navigating Manufacturing Complexity and the Economic Landscape of High-Cost Therapies
Despite the optimistic growth projections, the gene therapy market faces unique structural challenges, specifically regarding manufacturing scalability and pricing. The production of viral vectors is an intricate, multi-step process that demands stringent GMP compliance and specialized facilities, often leading to limited capacity and high per-unit costs. These manufacturing hurdles, combined with the extensive R&D required for rare disease indications, have resulted in premium pricing models that test the limits of current reimbursement frameworks. For example, Pfizer’s recently approved Beqvez for Hemophilia B represents the new wave of high-value, one-time treatments that require outcome-based pricing models to ensure patient access. To mitigate these restraints, major players like Biogen Inc., Sarepta Therapeutics, and Alnylam Pharmaceuticals are investing heavily in automation and AI-driven optimization to streamline the supply chain. While the high cost remains a barrier, the expansion into unmet medical needs and the rising global focus on genomic research, particularly in the fast-growing European market, provide a fertile ground for market players to implement flexible market-access strategies and secure long-term commercial success.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com