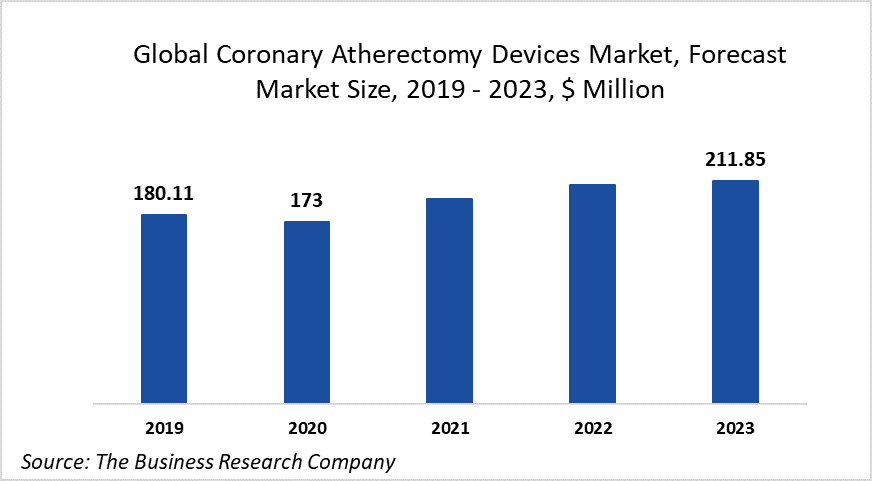
The global coronary atherectomy devices market 2020 is expected to decline from $180.11 million in 2019 to $173.32 million in 2020 at a compound annual growth rate (CAGR) of -3.77%. The decline is mainly due to the COVID-19 outbreak that has led to patients’ preference for deferred care to avoid visiting hospitals, which resulted in reduced patient volume. The coronary atherectomy equipment market is then expected to recover and reach $211.85 million in 2023 at a CAGR of 6.92%.
The increasing prevalence of cardiovascular diseases (CVDs) has resulted in an increased demand for atherectomy devices for the treatment of CVDs. The increased prevalence of cardiovascular diseases can be attributed to the use of smoking, physical inactivity, and unhealthy diet. For instance, according to the World Health Organization, in 2019, 17.9 million people died from CVDs worldwide every year. CVDs is a group of disorders of the heart and blood vessels which includes cerebrovascular disease, rheumatic heart disease, coronary heart disease, and other conditions. According to the British Heart Foundation, 2020, Coronary Heart Disease is responsible for around 64,000 deaths in the UK each year, an average of 180 people each day, or one death around every eight minutes. One in seven men and one in twelve women in the United Kingdom die from coronary heart disease. Therefore, increasing prevalence of cardiovascular diseases is expected to drive the growth of the global coronary atherectomy devices market.
Major players in the global coronary atherectomy devices market include Boston Scientific, Philips, Cardiovascular Systems, Spectranetics, Medtronic, Avinger, BIOTRONIK, Arterial Remodeling Technologies, Atrium Medical, B. Braun Melsungen, Balton, and Biosensors International. Large coronary atherectomy equipment market manufacturers are strategically partnering and collaborating with start-ups and mid-sized companies to broaden products and services. For instance, in July 2018, Cardiovascular Systems signed an agreement with OrbusNeich for selling its coronary and peripheral Orbital Atherectomy Systems (OAS) in other countries. In the same month, the company collaborated with Aerolase Corporation for developing a new laser atherectomy device for physicians, which can be used more effectively in the treatment of multiple forms of arterial disease.
Similarly, in May 2019, Cardiovascular Systems, Inc. acquired the WIRION Embolic Protection System and its related assets from Gardia Medical for $5.6 million. The WIRION System has higher adaptability than other available embolic protection systems because it can be used with any .014 guidewire and for all types of peripheral interventions. Also, the WIRION System is the only embolic protection device indicated for use with any atherectomy system. Gardia Medical Ltd is an Israel-based medical device company that produces specialized catheter-based delivery systems to deliver, lock, and use devices on any guidewire.
The coronary atherectomy devices market consists of sales of coronary atherectomy devices and related services by entities (organizations, sole traders and partnerships) that manufacture coronary atherectomy devices. Coronary atherectomy devices are used to remove atherosclerosis from coronary blood vessels.
Here Is A List Of Similar Reports By The Business Research Company:
Cardiovascular Surgery Devices And Equipment Global Market Report 2020
Cardiac Assist Devices And Equipment Global Market Report 2020
Cardiovascular Monitoring And Diagnostic Devices And Equipment Global Market Report 2020
Interventional Cardiology Devices And Equipment Global Market Report 2020
Interested to know more about The Business Research Company?
The Business Research Company is a market intelligence firm that excels in company, market, and consumer research. Located globally it has specialist consultants in a wide range of industries including manufacturing, healthcare, financial services, chemicals, and technology.
Media Contact
Company Name: The Business Research Company
Contact Person: Oliver Guirdham
Email: Send Email
Phone: +44 20 7193 0708
Address:
City: London
State: Greater London
Country: United Kingdom
Website: https://www.thebusinessresearchcompany.com/