CAR T Cell Therapy Market Analysis
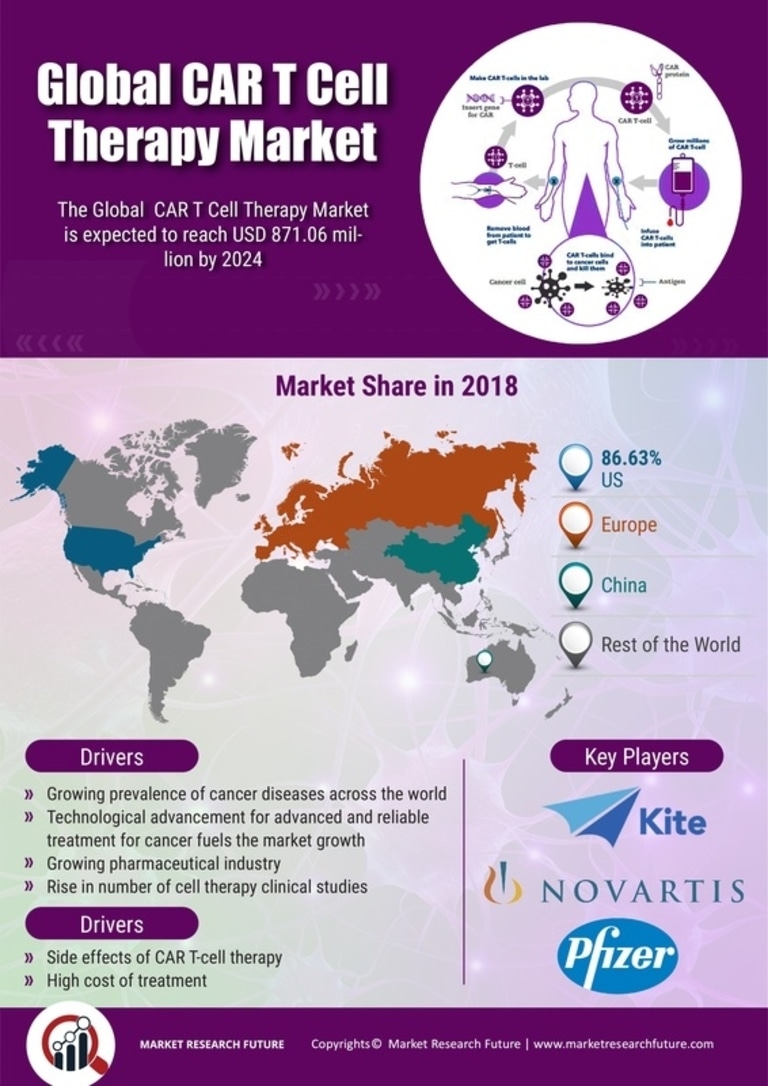
Global CAR-T cell therapy market is predicted to touch USD 8716.06 million at a 58.52% CAGR over the forecast period (2017-2025), in accordance with the latest Market Research Future (MRFR) report. CAR-T cell therapy, simply put, is a form of personalized immunotherapy which uses the power of the immune system of the body in fighting cancer. This is a noteworthy breakthrough in cancer treatment having a favorable impact on blood cancer patients. They are receptor proteins which have been engineered to provide T cells the ability to target a specific protein. These receptors are chimeric as they combine T-cell activating functions and antigen-binding into one receptor.
Various factors are propelling the CAR-T cell therapy market growth. These factors, as stated by the latest MRFR report, include increasing incidences of cancer worldwide, advancement in technology for reliable and advanced treatment for cancer, growing number of cell therapy clinical studies, and the growing number of patients showing response failure towards alternative treatments. Additional factors pushing market growth include growing initiatives by the government to promote research for cell therapy in cancer and improving health care infrastructure, and development of effective and new therapy options for curing different symptoms of the disease.
On the contrary, high treatment cost and side effects of CAR-T cell therapy are factors that may impede the CAR-T cell therapy market growth over the forecast period.
Avail Free Sample Copy at https://www.marketresearchfuture.com/sample_request/8102
CAR T Cell Therapy Market Segmentation
The MRFR report provides a complete segmental analysis of the CAR-T cell therapy market report on the basis of target antigen and application.
Based on target antigen, the CAR-T cell therapy market is segmented into CD22, CD19, and others. Of these, the CD19 target antigen segment will have the largest share in the market over the forecast period.
Based on application, the CAR-T cell therapy market is segmented into diffuse large B-cell lymphoma, acute lymphoblastic leukemia, and others.
CAR T Cell Therapy Market Regional Analysis
By region, the CAR-T cell therapy market report covers the latest trends and growth opportunities across Europe, the Asia Pacific (APAC), North America, and the Rest of the World (RoW). Of these, North America will spearhead the market over the forecast period. This can be chiefly attributed to factors such as growing awareness among people about treatment options and increasing incidences of cancer in this region. The US is the key contributor in this region owing to the growing number of cancer cases in this country. Moreover, high investment in research and development activities, presence of key players in the region, increase in healthcare expenditure, high availability to advanced therapies, and presence of advanced healthcare infrastructure is also boosting the market growth. Besides, favorable reimbursement policies and various government initiatives are also adding to the growth of the market in the region.
The CAR-T cell therapy market in Europe is predicted to have a healthy growth over the forecast period. This is on account of the rising incidences of hematologic cancer such as multiple myeloma, lymphoma, and leukemia. China is the chief contributor in this region owing to increasing disposable income and developing healthcare infrastructure.
The CAR-T cell therapy market in the APAC region is predicted to have a notable growth over the forecast period due to affordability and limited healthcare access.
CAR T Cell Therapy Market Key Players
Leading players profiled in the CAR-T cell therapy market report include Mustang Bio (US), Legend Biotech (US), Sorrento Therapeutics (US), Juno Therapeutics (US), CARsgen Therapeutics (China), Autolus Therapeutics PLC (UK), Cellectis (France), Kite Pharma (US), Pfizer, Inc. (US), and Novartis AG (Switzerland), among others. To improve their presence and increase user base in the market, key players are increasingly introducing innovative products, have a strong pipeline, and growing FDA approvals. Besides, they are also adopting various strategies for standing out as strong competitors as well as expanding their sales in the market. Some of these strategies include collaborations, geographic expansion, and acquisitions & merger policies.
CAR T Cell Therapy Industry News
September 2019: CT053 has received orphan drug status from the FDA. This is an investigational chimeric antigen receptor T-cell therapy to treat multiple myeloma. CARsgen Therapeutics or CT053 is a fully human, autologous CAR T-cell therapy which will target the B-cell maturation antigen on cancer cells’ surface.
Browse Complete Report with TOC at https://www.marketresearchfuture.com/reports/car-t-cell-therapy-market-8102
About Market Research Future:
At Market Research Future (MRFR), we enable our customers to unravel the complexity of various industries through our Cooked Research Report (CRR), Half-Cooked Research Reports (HCRR), Raw Research Reports (3R), Continuous-Feed Research (CFR), and Market Research & Consulting Services.
MRFR team have supreme objective to provide the optimum quality market research and intelligence services to our clients. Our market research studies by Components, Application, Logistics and market players for global, regional, and country level market segments, enable our clients to see more, know more, and do more, which help to answer all their most important questions.
In order to stay updated with technology and work process of the industry, MRFR often plans & conducts meet with the industry experts and industrial visits for its research analyst members.
Media Contact
Company Name: Market Research Future
Contact Person: Abhishek Sawant
Email: Send Email
Phone: +1 646 845 9312
Address:Market Research Future Office No. 528, Amanora Chambers Magarpatta Road, Hadapsar
City: Pune
State: Maharashtra
Country: India
Website: https://www.marketresearchfuture.com/reports/car-t-cell-therapy-market-8102