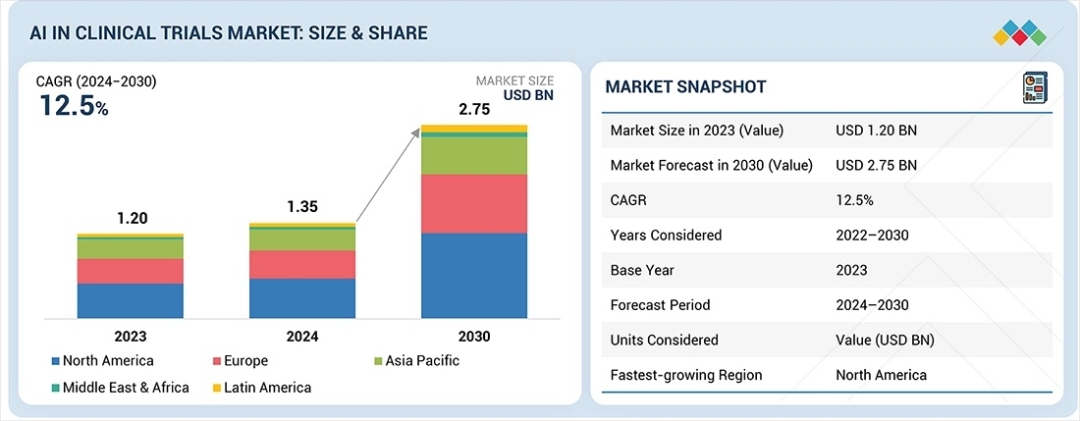
The global AI in clinical trials market is poised for significant expansion, with valuations projected to rise from USD 1.35 billion in 2024 to USD 2.75 billion by 2030. This growth, representing a compound annual growth rate (CAGR) of 12.5%, is primarily fueled by the urgent need within the pharmaceutical and biopharmaceutical sectors to reduce the staggering costs and extended timelines associated with traditional drug development. As clinical trials grow in complexity-particularly in specialized fields like oncology and rare diseases-AI-enabled solutions are becoming indispensable for maintaining data integrity, optimizing site selection, and enhancing patient recruitment strategies.
Download PDF Brochure:https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=42687548
Revolutionizing Patient Recruitment and Trial Design Efficiency
One of the most significant bottlenecks in modern drug development is the identification and retention of eligible trial participants. In 2023, the patient recruitment segment accounted for the largest share of the market, as sponsors increasingly turned to AI to parse through electronic health records (EHRs), genomic data, and real-world evidence. By utilizing advanced algorithms, researchers can match patients to specific trial criteria with unprecedented accuracy, effectively slashing recruitment timelines that have historically delayed drug launches by years.
Beyond recruitment, AI is fundamentally altering the architecture of trial protocols. Through predictive modeling and trial design optimization, biopharmaceutical companies can now simulate various study scenarios to identify potential risks before a single patient is enrolled. This proactive approach allows for the creation of adaptive trial models and the integration of virtual control arms, which utilize historical data to reduce the number of patients required for a study. These innovations not only lower operational risks but also support the industry’s shift toward personalized medicine, where treatments are tailored to specific genetic profiles and biomarkers.
Technological Dominance of Machine Learning and Cloud Integration
From a technical perspective, machine learning (ML) remains the dominant force within the market. ML algorithms are uniquely capable of analyzing the massive, unstructured datasets generated during clinical research, providing real-time insights that human clinicians simply cannot achieve at scale. Whether it is predicting treatment responses in Phase II clinical trials-a segment that currently dominates the market due to its high-stakes decision-making-or detecting subtle adverse events in post-market surveillance, machine learning provides the analytical backbone for the next generation of eClinical solutions.
This technological surge is further amplified by the rapid transition to cloud-based deployment. The industry is moving away from rigid, on-premises legacy systems in favor of scalable cloud platforms that facilitate global collaboration and decentralized trial models. Cloud integration allows for seamless data flow between Contract Research Organizations (CROs), hospitals, and sponsors, ensuring that data is accessible and secure regardless of geographic location. This flexibility is particularly vital in the wake of digital transformation initiatives in North America, which held a 41.6% revenue share in 2023 and continues to lead the global market in AI adoption.
Strategic Landscape and the Future of Oncology Trials
The competitive ecosystem is characterized by a mix of established “star” players and agile startups. Industry leaders such as IQVIA Inc., Dassault Systèmes (Medidata), and Insilico Medicine are setting the standard for end-to-end solutions that integrate everything from site optimization to regulatory compliance. Meanwhile, startups like Unlearn.ai and ReviveMed are carving out niches in digital twin technology and specialized metabolomics, respectively. Recent developments, such as the expanded partnership between Bioforum and Medidata in late 2024, underscore the industry’s commitment to improving data flow and compliance through AI-powered clinical data studios.
Request Sample Pages-https://www.marketsandmarkets.com/requestsampleNew.asp?id=42687548
Looking ahead to 2030, the oncology segment is expected to remain the dominant indication for AI application. The rising global burden of cancer, coupled with the intricate nature of targeted immunotherapies, necessitates the level of precision that only AI can provide. By identifying complex biomarkers and predicting how different patient subgroups will react to novel therapies, AI is not just an efficiency tool-it is a life-saving necessity. Despite challenges such as algorithm bias and the need for skilled AI professionals, the trajectory for AI in clinical trials is clear: it is the primary engine that will drive the future of medical innovation and the delivery of precision healthcare to patients worldwide.
Media Contact
Company Name: MarketsandMarkets™ Research Private Ltd.
Contact Person: Mr. Rohan Salgarkar
Email: Send Email
Phone: 18886006441
Address:1615 South Congress Ave. Suite 103, Delray Beach, FL 33445
City: Florida
State: Florida
Country: United States
Website: https://www.marketsandmarkets.com